Katherine Neil , Allison Marcil , Lynette Kosar , Zack Dumont , Lisa Ruda , Kaitlyn McMillanABSTRACT
Background:
Opioid analgesics are high-alert medications known to cause adverse drug events.
Objectives:
The purpose of this study was to determine the cause of opioid incidents requiring administration of naloxone, an opioid reversal agent. The specific objectives were to determine the number of opioid incidents and the proportion of incidents documented through occurrence reporting and to characterize the incidents by phase in the medication-use process, by type of incident, and by drug responsible for toxic effects.
Methods:
A retrospective chart analysis was conducted using records from 2 acute care centres in the Regina Qu’Appelle Health Region. The study included inpatients who received naloxone for reversal of opioid toxicity resulting from licit, in-hospital opioid use. Cases were classified as preventable or nonpreventable. Preventable cases were analyzed to determine the phase of the medication-use process during which the incident occurred. These cases were also grouped thematically by the type of incident. The drug most likely responsible for opioid toxicity was determined for each case. The proportion of cases documented by occurrence reporting was also noted.
Results:
Thirty-six cases involving administration of naloxone were identified, of which 29 (81%) were deemed preventable. Of these 29 preventable cases, the primary medication incident occurred most frequently in the prescribing phase (23 [79%]), but multiple phases were often involved. The cases were grouped into 6 themes according to the type of incident. Morphine was the drug that most frequently resulted in toxic effects (18 cases [50%]). Only two of the cases (5.6%) were documented by occurrence reports.
Conclusion:
Preventable opioid incidents occurred in the acute care centres under study. A combination of medication safety initiatives involving multiple disciplines may be required to decrease the incidence of these events and to better document their occurrence.
KEYWORDS: medication incident , medication safety , opioid , naloxone
RÉSUMÉ
Contexte :
Les analgésiques opioïdes sont des médicaments qui commandent une vigilance élevée, connus pour les événements indésirables qu’ils entraînent.
Objectifs :
Le but de cette étude était de déterminer la cause des incidents attribuables aux opioïdes nécessitant l’administration de naloxone, un antidote des opioïdes. Les objectifs précis étaient de déterminer le nombre d’incidents attribuables aux opioïdes et la proportion d’incidents constatés par comptes rendus d’événements et de caractériser les incidents selon la phase dans le processus de distribution des médicaments, le type d’incident et l’agent responsable des effets toxiques.
Méthodes :
Une analyse rétrospective des dossiers médicaux des patients a été menée dans deux centres de soins de courte durée de la Regina Qu’Appelle Health Region. L’analyse incluait les patients hospitalisés ayant reçu de la naloxone pour neutraliser la toxicité opioïde attribuable à l’utilisation intrahospitalière licite d’opioïdes. Les cas d’incidents ont été classés comme étant évitables ou non évitables. Les cas évitables ont été analysés afin de déterminer la phase du processus de distribution des médicaments durant laquelle l’incident est survenu. Ces cas ont également été regroupés par type d’incident. Le médicament le plus susceptible d’avoir causé une toxicité opioïde a été déterminé pour chaque cas. La proportion de cas constatés par comptes rendus d’événements a aussi été notée.
Résultats :
On a relevé 36 cas nécessitant l’administration de naloxone, dont 29 (81 %) ont été jugés évitables. De ces 29 cas évitables, le principal incident lié au médicament est survenu le plus souvent dans la phase de prescription (23 [79 %]), mais plusieurs phases étaient souvent en cause. Les cas ont été regroupés en six types d’incidents. La morphine était l’agent ayant le plus souvent entraîné des effets toxiques (18 cas [50 %]). Seulement deux des cas (5,6 %) ont été constatés par comptes rendus d’événements.
Conclusion :
Des incidents évitables liés aux opioïdes sont survenus dans les deux centres de soins de courte durée faisant l’objet de la présente analyse. Une combinaison de mesures faisant appel à plusieurs disciplines pour améliorer la sécurité des médicaments pourrait être nécessaire afin de réduire l’incidence de tels événements et de constater leur occurrence.
MOTS CLÉS: incident lié au médicament , sécurité des médicaments , opioïde , naloxone
[Traduction par l’éditeur]
Adverse events are a cause of morbidity and mortality among hospital patients worldwide, including Canada.1 The Canadian Adverse Events Study showed that 7.5% of hospital patients across Canada experienced an adverse event, and for 36.9% of these patients, the adverse events were preventable.2 The study also revealed that preventable adverse events caused significant harm to patients, resulting in death in some cases.2
An adverse drug event (ADE) is an adverse event involving medication. ADEs also contribute to morbidity and mortality, and some studies have suggested that the frequency of these events has been increasing over time.3,4 The Institute for Safe Medication Practices Canada (ISMP Canada) has defined an ADE as “an injury from a medicine or lack of an intended medicine”.5 Any ADE that is preventable is a medication incident.5
Certain medications are more likely to cause patient harm when involved in medication incidents. These medications are referred to as “high-alert medications”.5 To help raise awareness, ISMP Canada and its US counterpart, the Institute for Safe Medication Practices (ISMP) have developed lists of high-alert medications. In 1989, ISMP included morphine, an opioid analgesic, in its first list of high-alert medications.6 Since then, multiple studies have identified opioids as common causes of harmful and costly ADEs.7–10 ISMP and ISMP Canada also report medication incidents involving opioids, some of which have resulted in death.11,12 Three opioids (morphine, hydromorphone, and fentanyl) remain on ISMP Canada’s list of the top 5 drugs reported as causing harm through medication incidents, despite widespread education regarding the potential harms.13
When harmful opioid incidents occur, naloxone (an opioid receptor antagonist) can be used as an antidote to reverse the toxic effects. Given its direct relationship with meaningful, harmful opioid incidents, the administration of naloxone has been used in previous studies as a trigger tool to identify opioid incidents on a system-wide level.8,14,15 Use of trigger tools has been shown to be one of the most efficient and inclusive incident tracking methods available, and antidotes such as naloxone are the most specific markers within the trigger tool method.8,16 Thus, it is possible to use instances of naloxone administration as a method of identifying opioid incidents not identified through conventional incident tracking methods such as occurrence reporting.
The purpose of this study was to determine the cause of opioid incidents requiring the administration of naloxone in acute care centres to provide information supporting system improvements in the safe use of opioids. The frequency of opioid incidents was defined, and cases were characterized by phase of the medication-use process (i.e., prescribing, order entry and transcription, dispensing and delivery, administration, or monitoring), type of incident, and opioid involved. Each case was also assessed to determine if a corresponding occurrence report had been submitted.
This study used administration of naloxone as a marker to identify cases of opioid toxicity in 2 acute care centres in the Regina Qu’Appelle Health Region (total 639 beds). Cases that occurred during the 1-year period from December 1, 2009, to November 30, 2010, were reviewed. Ethics approval was obtained from the Regina Qu’Appelle Health Region Research Ethics Board.
Cases were included if naloxone had been administered to reverse opioid toxicity. Cases were excluded if toxicity was due to illicit drug use, if the opioids had been administered outside of the hospital, or if the patient was not admitted to hospital during or around the time of the event. Cases of naloxone administration for the treatment or prevention of bowel obstruction, urinary retention, or opioid-induced pruritus were also excluded. Cases were excluded if it was probable that the patient’s status was not due to opioid overdose, but rather to an underlying medical condition. This criterion was determined by a lack of objective or subjective improvement in respiratory distress, sedation, or hypotension after naloxone administration. Similar criteria have been applied in comparable studies.15,17 In some cases the principal investigator (K.N.) was unsure whether the patient’s clinical status before and after naloxone administration was indicative of opioid toxicity. In these instances, an additional investigator (L.K.) with more clinical experience in the area of pain management was consulted.
During the study timeframe, the majority of naloxone orders were entered into the pharmacy’s computerized order entry system. Computer system reports were therefore used as the primary method of identifying possible instances of naloxone administration. However, these reports included cases in which an order was written proactively but the drug was never administered. As it was not possible to examine all of the charts, the reports were compared with ward stock requests for restocking naloxone to refine the list of patients who may have required the reversal agent. Occurrence reports and “code blue” records were also reviewed to identify additional naloxone orders that may not have been entered into the computer system.
The following information was collected from patients’ charts: demographic characteristics, symptoms indicating opioid toxicity, naloxone regimen and its effect, and opioid orders and administration. Disease states and conditions affecting susceptibility to opioid toxicity (e.g., kidney or liver dysfunction, extremes of age) were also noted.
Cases of naloxone administration that met the inclusion criteria were analyzed independently by a panel comprising the principal investigator (K.N.), who was a pharmacy resident at the time, and 2 experienced hospital pharmacists (L.K. and A.M.). The panelists determined whether each case was preventable or nonpreventable. Each preventable case was assessed to determine the phase or phases of the medication-use process involved (prescribing, order entry and transcription, dispensing and delivery, administration, monitoring, or other). The panelists first determined which phase was primarily responsible for the medication incident and resultant naloxone administration (the primary phase). The panelists also established if other phases of the medication-use process had contributed to the incident; for example, an incident might have occurred during the prescribing phase, but might have been overlooked during the order entry or administration phase. Ratings were based on a majority, i.e., 2 of the 3 raters having the same independent assessment. If the 3 panelists assessed a case differently, that case was discussed jointly until a consensus was reached. To determine the validity of this method, the individual ratings were compared to the overall analysis using Cohen’s kappa to correct for chance agreement. To assess whether the distribution of primary phase assessments was significantly different than what would have been expected by chance, a one-sample chi-squared test was performed (with nonpreventable cases excluded).
The principal investigator grouped the preventable cases thematically according to the type of incident, which was determined by examining the circumstances and contributing factors surrounding the incident. The principal investigator examined all cases (preventable and nonpreventable) to determine the drug and route of administration most responsible for the ADE. Finally, every case was assessed to determine if it had been documented by occurrence reporting.
A total of 512 charts were screened, and 36 cases met the inclusion criteria. Twenty (56%) of the patients were male, and the mean age was 65.6 years (range 0–93 years). In 23 (64%) of the cases, respiratory depression and sedation were observed. Sedation without respiratory depression was observed in 10 cases (28%), and respiratory depression alone accounted for 2 cases (5.6%). Additional symptoms such as hypotension, diaphoresis, meiosis, and nausea were noted in 10 cases. In one case, no symptoms were documented; however, opioid toxicity was strongly suspected, as the incident occurred during a gastroscopy and colonoscopy procedure, and the patient was returned to the unit in a drowsy state soon after naloxone was administered. The total dose of naloxone ranged from 0.04 mg to 6 mg. The regimens consisted of between 1 and 8 doses, with individual doses ranging from 0.04 mg to 2 mg. In addition, 2 patients required continuous infusion of naloxone over several hours. Two (5.6%) of the cases were documented through occurrence reporting.
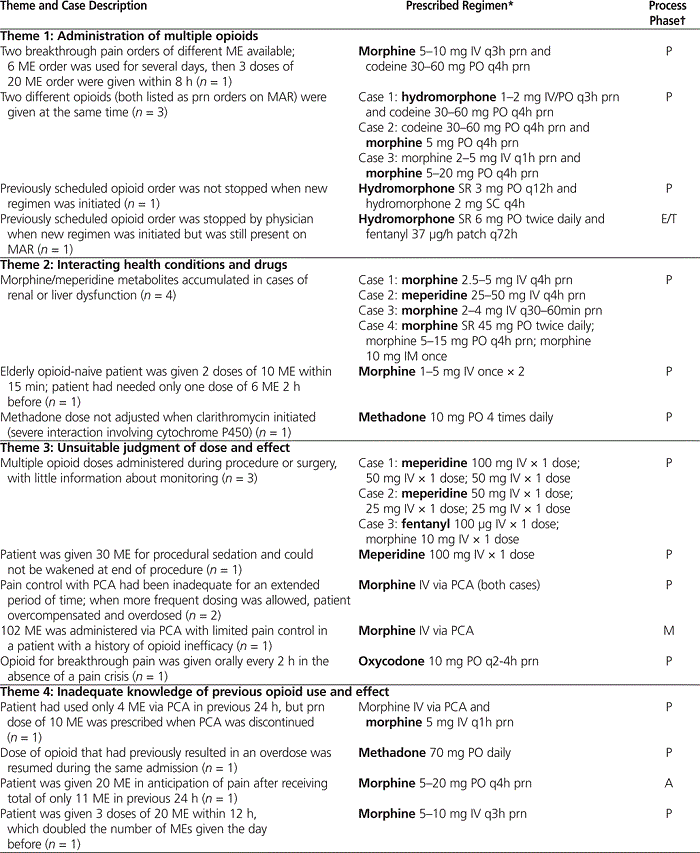
Seven cases (19%) were coded as “nonpreventable,” indicating that an ADE had occurred in the absence of a medication incident. Twenty-nine cases (81%) were determined to have been preventable medication incidents. Among the preventable cases, the majority of medication incidents were coded as occurring in the prescribing phase of the medication-use process ( n = 23 [79%]), significantly more than occurred in the other phases ( p = 0.002). The primary phase of the medication process that contributed to each preventable case, as well as a summary and description of cases, is shown in Table 1.
Table 1.
Summary of Preventable Cases Grouped by Thematic Coding


The panelists indicated that in over half of the preventable cases ( n = 17 [59%]), multiple phases of the medication process had contributed to naloxone administration. Prescribing was the primary phase contributing to naloxone administration in the majority of preventable cases (23 [79%]) (Table 2).
Table 2.
Phase of the Medication-Use Process Contributing to Preventable Opioid Incidents*
Cohen’s kappa was used to assess the agreement between pharmacists’ independent assessments of preventability and the primary phase of the medication-use process involved. Significant agreement was found between the final overall assessment and all assessors. Kappa was significant for the primary investigator (K = 0.41, z = 5.14, p < 0.001), pharmacist 2 (K = 0.82, z = 8.19, p < 0.001), and pharmacist 3 (K = 0.58, z = 5.59, p < 0.001).
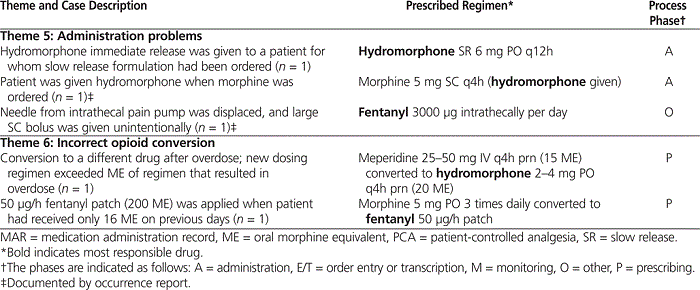
The 29 preventable cases were grouped into 6 themes on the basis of the type of incident that had occurred and the surrounding circumstances (see Table 1).
The first thematic grouping consisted of 6 cases involving administration of multiple opioids. This theme included cases in which 2 different opioids for breakthrough pain or 2 different scheduled opioids were administered at the same time. It also included a case in which 2 opioids of different potency were available for administration, with administration of multiple doses of the stronger opioid resulting in overdose.
The second theme accounted for another 6 cases, which were determined to have occurred because of interactions between the opioid administered and patients’ health conditions or other drugs. Some cases within this theme were related to renal or hepatic dysfunction. Cases exhibiting the effects of advanced age and a pharmacokinetic drug interaction were also included under this theme.
The third theme comprised 8 cases that occurred because of an unsuitable judgment of dose and effect. This group included instances in which the patient required naloxone after a procedure or after surgery, as well as one case in which the frequency of dosing was not appropriate for the type of pain being treated. Cases involving inadequate monitoring of pain control while using patient-controlled analgesia were also included within this theme.
Four cases were determined to have been due to inadequate knowledge of the patient’s previous opioid use and effects. This theme included cases in which the practitioner made changes to an opioid regimen or administered a dose from the upper end of a dosing range without considering the patient’s previous needs for and effects of opioids.
The fifth thematic grouping comprised 3 cases that occurred because of administration problems. These incidents involved administration of medications that were different from those ordered by the physician, as well as one case involving a medication pump (although it is unknown whether the source of the incident was operator error or mechanical failure). The 2 cases included within the sixth theme occurred because of incorrect conversions when a patient’s regimen was changed from one opioid to another.
Examination of all 36 preventable and nonpreventable cases showed that morphine contributed to half of all ADEs ( n = 18 [50%]) (Table 3). The majority of medications causing ADEs were administered intravenously ( n = 21 [58%]) (Table 4).
Table 3.
Primary Drug Linked to Toxicity (All Cases*)
Table 4.
Primary Route of Administration Linked to Toxicity (All Cases*)
This investigation has shown that ADEs involving the licit use of opioid analgesics do occur among inpatients. Over the 1-year period of the study, 36 cases of opioid-related ADEs occurred in 2 acute care centres (total 639 beds). A study with similar methodology produced comparable results, with 22 cases being identified in 2 acute care centres (total 619 beds).15
The majority of preventable opioid incidents identified in this investigation occurred in the prescribing phase of the medication-use process ( n = 23 [79%]). Two previous studies in different health care settings yielded similar results, with the prescribing phase involved in 49% and 97% of instances, respectively.9,18 These studies relied on a combination of chart review, occurrence reporting, and surveys of patients and staff. Conversely, studies that used occurrence reporting as the primary method of incident detection have described medication incidents most commonly occurring in the administration phase.19,20 It is possible that this variation in results is related to the method of identifying incidents. A previous survey indicated that the majority of nurses submit occurrence reports regarding their own errors more than 80% of the time, whereas the majority of physicians complete reports for less than 20% of their own incidents.21 This disparity could account for the large quantity of administration errors identified in studies that rely on occurrence reports alone.
This study did not identify any medication incidents occurring in the dispensing or delivery phase of the medication process. However, previous literature has suggested that medication incidents do occur during this phase.9,18,19 It is possible that this type of incident was not identified because of the small number of cases uncovered. It is also conceivable that dispensing and delivery errors do not commonly result in incidents severe enough to require naloxone or that these incidents are identified and rectified during the administration phase and never reach the patient.
Although prescribing was the phase of the medication-use process most commonly associated with incidents, many of the cases in this investigation resulted from incidents involving multiple phases. This finding speaks to the complexity of the cases identified, as well as the shared responsibility of health care providers to prevent opioid incidents. It also suggests that efforts to make opioid use safer should involve all health care providers.
The thematic analysis in this investigation provides valuable information suggesting targeted changes that could be implemented in acute care centres to decrease opioid incidents (Table 5). These recommendations have been ranked, using ISMP Canada’s “hierarchy of effectiveness”,22 according to how likely they are to be effective in achieving safe outcomes. The hierarchy lists the following actions in order of increasing effectiveness: education and provision of information; rules and policies; reminders, checklists, and double checks; simplification and standardization; automation and computerization; and forcing functions.22
Table 5.
Summary of Study Findings and Recommendations
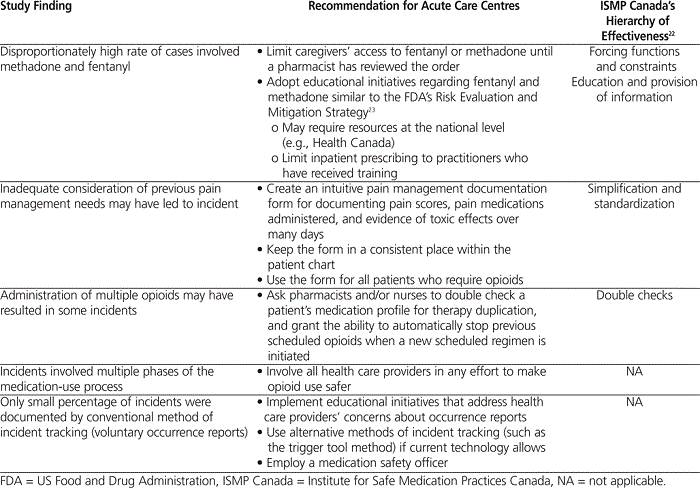
In this investigation, morphine, hydromorphone, and IV meperidine were the drugs most frequently associated with ADEs. This result is not surprising, as these drugs accounted for the majority of total opioid orders prescribed in the acute care centres during the study period (see Table 3). For other opioids, such as fentanyl and methadone, there was a larger disparity between the percentage of incidents uncovered in this investigation and the percentage of total orders written in the hospital (see Table 3). The literature is rich with case reports and articles describing harm due to the pharmacokinetic and administration challenges of fentanyl and methadone.24–28 In response, ISMP and ISMP Canada have published multiple strategies to manage these medications safely. One strategy involves limiting access to fentanyl and methadone in patient care areas, to ensure that a pharmacist is able to review all orders for these medications before administration.26,28 With the emergence of controlled-access cabinets in Canadian acute care centres, it may be possible to keep these important medications in patient care areas, yet allow the pharmacist to regulate their release to caregivers. This approach is an example of a forcing function, according to ISMP Canada’s hierarchy of effectiveness, as it makes application of fentanyl patches impossible without a pharmacist’s review. This action could be expected to be quite successful in improving patient safety, as it is the highest ranked type of action in the hierarchy. An additional management strategy that ISMP and ISMP Canada have proposed is ensuring that practitioners have received adequate training regarding methadone and fentanyl.24,28 The US Food and Drug Administration has echoed this recommendation through the creation of an approved Risk Evaluation and Mitigation Strategy (REMS) targeting long-acting opioid products, including methadone and transdermal fentanyl.23 This strategy strongly encourages practitioners who prescribe long-acting opioids to educate themselves about the complexities and consequences of these products by consulting the REMS-compliant educational material provided by the manufacturer.23 The creation of similar initiatives through Health Canada may decrease opioid incidents in Canada. Although educational initiatives might be expected to have limited effectiveness, given the position of education and information in ISMP Canada’s hierarchy of effectiveness, they may still have value, given that some of the educational points required in REMS-compliant materials (such as awareness of drug interactions and opioid conversions) relate to incidents identified in this study. Limiting inpatient prescribing of transdermal fentanyl patches to physicians who have been adequately trained (as has been done with methadone) may decrease future incidents involving fentanyl.11
Some cases of opioid toxicity analyzed in this study might have been prevented if the health care providers had had a better understanding of the patient’s previous opioid use and pain management needs. A more user-friendly pain management documentation form, clearly indicating all pain medications administered and their effects over several days, could be developed and used to mitigate this problem. Consistent use and placement of this form in the charts of all patients receiving opioids could potentially make monitoring of previous opioid requirements and response simpler and could also decrease the aforementioned issue of duplicate therapy. This standardization of pain control documentation could prove successful, as standardization is one of the more effective actions in the hierarchy of effectiveness. An environmental scan of pain management forms used by other health regions might assist in the creation of an intuitive method of documentation.
A large proportion of cases in this study resulted from administration of multiple opioids. Asking pharmacists or nurses to review patient medication profiles and granting them the ability to automatically stop scheduled opioids when a new scheduled regimen is initiated might decrease the duplication of opioids that could otherwise result in harm to patients. This double check of the medication profile could yield moderate improvements in patient safety, according to the ISMP Canada hierarchy of effectiveness.
Only 2 (5.6%) of the 36 cases included in this investigation were documented by occurrence reports. This degree of overlap is in keeping with a recent systematic review of incident identification methods, which reported that trigger tools and occurrence reports identified the same incident in only 0.5%–10% of cases.16 The review went on to reveal that occurrence reporting is the method least likely to identify drug-related problems.16 Given that occurrence reporting is currently the only incident tracking method available in the acute care centres where this study took place, a more effective process is needed. In previous surveys, reasons for under-reporting among health care providers were uncertainty about what should be reported and who should do the reporting, as well as concerns about implicating colleagues, legal ramifications, and time constraints.21,29 Health care providers reported that they would be more likely to submit occurrence reports if they could receive regular feedback about the reported incidents and if they believed or witnessed that occurrence reporting changed patient care and system processes.30 These providers would also be more likely to report if they could submit in an electronic format or if they were to receive further education to help determine what type of incident requires a report.21 A previous initiative to address some of these issues significantly increased the number of occurrence reports submitted over a variety of hospitals and units.29 Acute care centres in Canada that wish to increase incident reporting may achieve a comparable increase through similar programming.
Other methods of incident identification could be explored. A recent systematic review reported the trigger tool method (used in this investigation) as the least labour-intensive and the least expensive of all conventional incident-tracking methods.16 However, the trigger tool method discussed in the systemic review was a fully operationalized, automated process. In hospitals without the technology necessary to track medication administration, a fully automated trigger tool system would not be possible. For acute care centres with widespread use of controlled-access cabinets (which record all drug withdrawals) or those that use electronic medication administration records, adoption of an automated trigger tool is possible.
The creation of a staff position dedicated to medication safety could improve incident reporting or even pave the way for alternative methods of incident tracking. The American Society of Health-System Pharmacists has described the training and employment of a medication safety officer, who would be responsible for increasing the reporting of ADEs, performing follow-up on reported occurrences, overseeing analysis of data, and assisting in development and implementation of process changes to improve medication safety.31
This study was limited by the retrospective nature of data collection. Clinical situations may have been interpreted incorrectly if charting was incomplete or vague. This investigation may have underestimated the incidence of harmful opioid incidents, as it was not possible to examine all charts because of time constraints. In addition, some incidents may not have been captured by the 3 methods used to identify naloxone administration. Furthermore, although the investigators established criteria to determine whether naloxone had alleviated the symptoms of opioid toxicity, they did not assess whether the patient’s clinical status was severe enough to warrant naloxone administration. Although the appropriateness of naloxone administration was not the focus of this study, this omission may have led to overestimation of the degree of patient harm experienced. The data analysis also had limitations. Pharmacodynamic drug interactions were not considered in this study because of their possible variability. If this type of interaction had been accounted for, the analysis might have yielded different results. There is also the risk that the results were skewed because of the homogenous nature of the analysis panel. In the future it would be beneficial to have a myriad of professionals involved, to stimulate a more thorough assessment of the cases, as well as to facilitate policy changes in a more holistic fashion.
Adverse drug events involving opioids occurred in the acute care centres under study. A combination of medication safety initiatives involving multiple disciplines may be required to decrease the incidence of these events, as well as to better document their occurrence.
1. Jha AK, Prasopa-Plaizier N, Larizgoitia I, Bates DW. Patient safety research: an overview of the global evidence. Qual Saf Health Care. 2010;19(1):42–7.
2. Baker GR, Norton PG, Flintoft V, Blais R, Brown A, Cox J, et al. The Canadian Adverse Events Study: the incidence of adverse events among hospital patients in Canada. CMAJ. 2004;170(11):1678–86.

3. Strausberg J, Hasford J. Drug-related admissions and hospital-acquired adverse drug events in Germany: a longitudinal analysis from 2003 to 2007 of ICD-10-coded routine data. BMC Health Serv Res. 2011;11:134.
4. Dequito AB, Mol PG, van Doormaal JE, Zaal RJ, van den Bemt PM, Haaijer-Ruskamp FM, et al. Preventable and non-preventable adverse drug events in hospitalized patients: a prospective chart review in the Netherlands. Drug Saf. 2011;34(11):1089–100.
5. Definitions of terms. Toronto (ON): Institute for Safe Medication Practices Canada; 2012 [cited 2012 Apr 25]. Available from: http://www.ismp-canada.org/definitions.htm
6. Davis NM, Cohen MR. Today’s poisons: how to keep them from killing your patients. Nursing. 1989;19(1):49–51.
7. Oderda GM, Said Q, Evans RS, Stoddard GJ, Lloyd J, Jackson K, et al. Opioid-related adverse drug events in surgical hospitalizations: impact on costs and length of stay. Ann Pharmacother. 2007;41(3):400–6.
8. Classen DC, Pestotnik SL, Evans RS, Burke JP. Computerized surveillance of adverse drug events in hospital patients. JAMA. 1991;266(20):2847–51.
9. Bates DW, Cullen DJ, Laird N, Petersen LA, Small SD, Servi D, et al. Incidence of adverse drug events and potential adverse drug events: implications for prevention. JAMA. 1995;274(1):29–34.
10. Kanjanarat P, Winterstein AG, Johns TE, Hatton RC, Gonzalez-Rothi R, Segal R. Nature of preventable adverse drug events in hospitals: a literature review. Am J Health Syst Pharm. 2003;60(17):1750–9.
11. Ongoing, preventable fatal events with fentanyl transdermal patches are alarming! ISMP Med Saf Alert. 2007 [cited 2012 Dec 23];12(13):1–3. Available from: http://www.ismp.org/newsletters/acutecare/articles/20070628.asp
12. Hydromorphone intended for an adult patient inadvertently administered to an infant. ISMP Can Saf Bull. 2008 [cited 2012 Dec 23];8(6):1–3. Available from: http://ismp-canada.org/download/safetyBulletins/ISMPCSB2008-06Hydromorphone.pdf
13. Top 10 drugs reported as causing harm through medication error. ISMP Can Saf Bull. 2006 [cited 2012 Dec 23];6(1):1–2. Available from: http://www.ismp-canada.org/download/safetyBulletins/ISMPCSB2006-01Top10.pdf
14. Rozich JD, Haraden CR, Resar RK. Adverse drug event trigger tool: a practical methodology for measuring medication related harm. Qual Saf Health Care. 2003;12(3):194–200.

15. Whipple JK, Ausman RK, Quebbeman EJ. Narcotic use in the hospital: reasonably safe? Ann Pharmacother. 1992;26(7–9):897–901.
16. Meyer-Massetti C, Cheng CM, Schwappach DL, Paulsen L, Ide B, Meier CR, et al. Systematic review of medication safety assessment methods. Am J Health Syst Pharm. 2011;68(3):227–40.
17. Whipple JK, Quebbeman EJ, Lewis KS, Gottlieb MS, Ausman RK. Difficulties in diagnosing narcotic overdoses in hospitalized patients. Ann Pharmacother. 1994;28(4):446–50.
18. Tam KW, Kwok KH, Fan YM, Tsui KB, Ng KK, Ho KY, et al. Detection and prevention of medication misadventures in general practice. Int J Qual Health Care. 2008;20(3):192–9.
19. Cornish P, Etchells E, Kahan M, Salsman B, Sawyer J, Senders J. Shared learning—reported incidents involving hydromorphone. ISMP Can Saf Bull. 2006;6(9):1–3. Available from: http://www.ismp-canada.org/download/safetyBulletins/ISMPCSB2006-09Hydromorphone.pdf
20. McDonnell C. Opioid medication errors in pediatric practice: four years’ experience of voluntary safety reporting. Pain Res Manage. 2011;16(2):93–8.
21. Taylor JA, Brownstein D, Christakis DA, Blackburn S, Strandjord TP, Klein EJ, et al. Use of incident reports by physicians and nurses to document medical errors in pediatric patients. Pediatrics. 2004;114(3):729–35.
22. Greenall J, Senders JW. Medication safety alerts. Root cause analysis: learning from adverse events and near misses. Can J Hosp Pharm. 2006;59(1):34–6.
23. Extended-release (ER) and long-acting (LA) opioid analgesics risk evaluation and mitigation strategy (REMS). Silver Spring (MD): US Food and Drug Administration; 2012 [cited 2012 Aug 28]. Available from: http://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/UCM311290.pdf
24. Analysis of international findings from incidents involving fentanyl transdermal patches. ISMP Can Saf Bull. 2009 [cited 2012 Dec 23];9(10):1–2. Available from: http://ismp-canada.org/download/safetyBulletins/ISMPCSB2009-10-AnalysisOfInternationalFindingsFromIncidentsInvolvingFentanylTransdermal.pdf
25. Cheng R, Samples C, U D, Hyland S, Ho C. Fentanyl patches: analysis of international medication incidents [abstract]. Can J Hosp Pharm. 2010;63(4):342.
26. Fentanyl patch linked to another death in Canada. ISMP Can Saf Bull. 2007 [cited 2012 Dec 23];7(5):1–2. Available from: http://www.ismp-canada.org/download/safetyBulletins/ISMPCSB2007-05Fentanyl.pdf
27. Methadone: not your typical narcotic! ISMP Can Saf Bull. 2003 [cited 2012 Dec 23];3(12):1–2. Available from: http://ismp-canada.org/download/safetyBulletins/ISMPCSB2003-12Methadone.pdf
28. Keeping patients safe from iatrogenic methadone overdoses. ISMP Med Saf Alert. 2008 [cited 2012 Dec 23];13(3):1–3. Available from: https://http://www.ismp.org/newsletters/acutecare/articles/20080214.asp
29. Evans SM, Smith BJ, Esterman A, Runciman WB, Madderm G, Stead K, et al. Evaluation of an intervention aimed at improving voluntary incident reporting in hospitals. Qual Saf Health Care. 2007;16(3):169–75.

30. Katz R, Lagasse RS. Factors influencing the reporting of adverse perioperative outcomes to a quality management program. Anasth Analg. 2000;90(2): 344–50.
31. Medication safety officer sample job description. Bethesda (MD): American Society of Health-System Pharmacists; 2012 [cited 2012 May 17]. Available from: http://www.ashp.org/DocLibrary/Policy/PatientSafety/Sample-Job-Description-1.aspx
All funding for this project was provided by the Regina Qu’Appelle Health Region Department of Pharmacy Services, as a component of the Pharmacy Residency Program. We thank the Department of Risk Management and the Department of Health Information Management Services within the Regina Qu’Appelle Health Region for supporting data collection. We acknowledge Ali Bell for her assistance with the statistical analysis, Michael Stuber and Bill Semchuk for their contributions to idea generation and project development, and Corey Herod, Elsie Wong, and Doug Sellinger for information technology support.
Competing interests: None declared.
Canadian Journal of Hospital Pharmacy , VOLUME 66 , NUMBER 5 , September-October 2013