Francine Kwee, Sandra A N Walker, Marion Elligsen, Lesley Palmay, Andrew Simor, Nick DanemanABSTRACT
Background:
Pseudomonas aeruginosa, one of the leading causes of nosocomial gram-negative bloodstream infections, is particularly difficult to treat because of its multiple resistance mechanisms combined with a lack of novel antipseudomonal antibiotics. Despite knowledge of time-dependent killing with ß-lactam antibiotics, most hospitals in Canada currently administer ß-lactam antibiotics by intermittent rather than extended infusions.
Objectives:
To determine clinical outcomes, microbiological outcomes, total hospital costs, and infection-related costs for patients with P. aeruginosa bacteremia who received intermittent IV administration of antipseudomonal ß-lactam antibiotics in a tertiary care institution.
Methods:
For this retrospective descriptive study, data were collected for patients who were admitted between March 1, 2005, and March 31, 2013, who had P. aeruginosa bacteremia during their admission, and who received at least 72 h of treatment with ceftazidime, meropenem, or piperacillin–tazobactam. Clinical and microbiological outcomes were determined, and total and infection-related hospital costs were calculated.
Results:
A total of 103 patients were included in the analysis, of whom 79 (77%) experienced clinical cure. In addition, bacterial eradication was achieved in 41 (87%) of the 47 patients with evaluable data for this outcome. Twenty-eight (27%) of the 103 patients died within 30 days of discontinuation of antipseudomonal ß-lactam antibiotic therapy. The median total cost of the hospital stay was $121 718, and the median infection-related cost was $29 697.
Conclusions:
P. aeruginosa bacteremia is a clinically significant nosocomial infection that continues to cause considerable mortality and health care costs. To the authors’ knowledge, no previous studies have calculated total and infection-related hospital costs for treatment of P. aeruginosa bacteremia with intermittent infusion of antipseudomonal ß-lactam antibiotics, with characterization of cost according to site of acquisition of the infection. This study may provide important baseline data for assessing the impact of implementing extended-infusion ß-lactam therapy, antimicrobial stewardship, and infection control strategies targeting P. aeruginosa infection in hospitalized patients.
KEYWORDS: bacteremia, Pseudomonas, Pseudomonas aeruginosa, outcomes, ceftazidime, meropenem, piperacillin–tazobactam, intermittent infusion, infectious disease
RÉSUMÉ
Contexte :
La bactérie Pseudomonas aeruginosa, l’une des principales causes des bactériémies nosocomiales à Gram négatif, est particulièrement difficile à traiter, parce qu’elle possède de nombreux mécanismes de résistance et qu’il n’y a pas de nouveaux antibiotiques anti-Pseudomonas. Bien que l’on sache que l’effet bactéricide des ß-lactamines est fonction du temps, la plupart des hôpitaux au Canada administrent actuellement ces antibiotiques par perfusions intermittentes plutôt que prolongées.
Objectifs :
Déterminer quels sont les résultats cliniques, les résultats microbiologiques, les coûts totaux d’hospitalisation et ceux liés à l’infection seule pour les patients atteints d’une bactériémie à P. aeruginosa ayant reçu des ß-lactamines anti-Pseudomonas par perfusion intermittente dans un établissement de soins tertiaires.
Méthodes :
Pour la présente étude rétrospective descriptive, on a recueilli des données sur des patients hospitalisés entre le 1er mars 2005 et le 31 mars 2013, qui étaient atteints d’une bactériémie à Pseudomonas aeruginosa, et qui ont reçu au moins 72 heures de traitements par ceftazidime, méropenem ou pipéracilline-tazobactam. Les résultats cliniques et microbiologiques ont été déterminés, et les coûts totaux d’hospitalisation et ceux liés à l’infection ont été calculés.
Résultats :
En tout, 103 patients ont été retenus pour l’analyse. Parmi eux, 79 (77 %) ont joui d’une guérison clinique. De plus, une éradication bactérienne a été obtenue chez 41 (87 %) des 47 patients ayant des données évaluables pour ce résultat. Vingt-huit (27 %) des 103 patients sont décédés dans les trente jours suivant la fin de l’antibiothérapie par ß-lactamines anti-Pseudomonas. Le coût total médian du séjour à l’hôpital était de 121 718 $ et le coût médian lié à l’infection était de 29 697 $.
Conclusions :
La bactériémie à Pseudomonas aeruginosa est une infection nosocomiale cliniquement significative. Elle demeure à ce jour une cause importante de mortalité et une source considérable de coûts de soins de santé. À la connaissance des auteurs, aucune étude antérieure n’a calculé les coûts totaux d’hospitalisation et ceux liés à l’infection pour le traitement de la bactériémie à Pseudomonas aeruginosa par perfusion intermittente de ß-lactamines anti-Pseudomonas, en caractérisant ces coûts selon le site de transmission de l’infection. Cette étude pourrait fournir d’importantes données de référence pour évaluer l’effet de la mise en place du traitement par perfusion prolongée de ß-lactamines, d’une gestion responsable des antimicrobiens et de stratégies de prévention des infections qui visent l’infection à P. aeruginosa chez les patients hospitalisés.
MOTS CLÉS: bactériémie, Pseudomonas, Pseudomonas aeruginosa, résultats, ceftazidime, méropénem, pipéracilline-tazobactam, perfusion intermittente, maladie infectieuse
Pseudomonas aeruginosa is a major nosocomial and prototypically antibiotic-resistant pathogen that can cause serious infections, especially in immunocompromised and critically ill patients.1–3 It is present in about 4% of all cases of nosocomial infection with positive blood culture results, with reported mortality rates ranging from 18% to 61%.4–7 ß-Lactam antibiotics exhibit time-dependent bactericidal activity, meaning that their efficacy is optimized by maximizing the time that free-drug concentration is above the minimum inhibitory concentration (MIC) (%f T>MIC).8 This characteristic is currently not being fully exploited, as many hospitals continue to manage these potentially severe infections with intermittent (rather than extended) IV infusions of ß-lactam antibiotics. With intermittent dosing, ß-lactams quickly attain a high peak concentration, followed by a rapid drop in serum drug concentration as a result of the short half-life. Attaining a targeted %f T>MIC of at least 50% with intermittent ß-lactam dosing may be particularly difficult for pathogens such as P. aeruginosa because of potentially elevated MICs.9 Recognizing the importance of maximizing the efficacy of currently available antimicrobial agents, the Infectious Diseases Society of America and the Society of Healthcare Epidemiology of America have recognized extended infusion of ß-lactams as an appropriate method for optimizing the use of these important antibiotics.10
Although strong pharmacodynamic data suggest that extended-infusion dosing may be more effective than intermittent dosing, it is unclear whether this approach translates to improved patient outcomes, because there is a lack of appropriately powered prospective randomized comparative studies.11 The primary objective of this study was to characterize the microbiological outcomes, clinical outcomes, and costs (both total hospital costs and infection-related costs) for patients with P. aeruginosa bacteremia treated with intermittent IV infusion of ß-lactams (ceftazidime, meropenem, or piperacillin–tazobactam). The secondary objective was to determine the necessary sample size and therefore the feasibility of performing a prospective study to compare extended and intermittent infusion regimens in a single centre.
This retrospective descriptive study was performed at a tertiary care teaching hospital in Toronto, Ontario. The study was approved by the Sunnybrook Health Sciences Centre Research Ethics Board on February 7, 2013. Electronic hospital databases were used to identify adult inpatients (≥ 18 years of age) admitted between March 1, 2005, and March 31, 2013, who had positive results on blood culture for Pseudomonas aeruginosa and who were treated with ceftazidime, meropenem, or piperacillin–tazobactam. Patients with polymicrobial bacteremia and patients who received less than 72 h of antipseudomonal ß-lactam therapy were excluded from assessment. Relevant data were obtained from the patients’ medical charts and from electronic patient records by a single reviewer (F.K.). Patient-specific hospital costs were obtained from the hospital’s Business Office.
Data collected were age, sex, hospital location at time of bacteremia diagnosis (ward or intensive care unit), underlying diseases, length of stay, duration of hospital stay before collection of samples for culture, interval between culture date and initiation of ß-lactam antipseudomonal therapy, focus of infection, site of acquisition of the infection (nosocomial, health care–associated, or community-acquired), history of an invasive procedure within 72 h before collection of samples for culture, antibiotic use within 3 months of positive blood culture result, presence of an intravascular venous catheter (i.e., central line, arterial catheter, peripheral IV catheter), presence of a urinary catheter, use of mechanical ventilation or hemodialysis during the hospital stay before the incident culture date, serum creatinine, neutrophil count, Charlson comorbidity index,12 Pitt bacteremia score13 on the day of blood sampling for culture, and information about the antibiotic regimens prescribed for treatment of P. aeruginosa bacteremia.
P. aeruginosa bacteremia was defined as identification of the organism from one or more sets of blood culture samples. For any patient, only the first episode of P. aeruginosa bacteremia during a single hospital admission was assessed. All cultured isolates were deemed clinically significant and were therefore tested for antimicrobial susceptibility. ß-Lactam antipseudomonal therapy was considered to be definitive therapy if the antibiotic was administered for at least 72 h and was started no more than 72 h before or up to 7 days after collection of blood samples for culture. Each case of bacteremia was categorized, according to acquisition site, as nosocomial, health care–associated, or community-acquired.14 Cases were defined as having nosocomial acquisition of P. aeruginosa bacteremia if the first blood sample with a positive culture result for P. aeruginosa was drawn following a hospital stay of at least 48 h. Cases of health care–associated bacteremia were defined as those in which the first blood sample with positive culture result for P. aeruginosa was drawn less than 48 h after hospital admission in patients who fulfilled any of the following criteria: (1) had received IV therapy at home, wound care, or specialized nursing care through a health care agency, family, or friends, or self-administered IV medical therapy within 30 days before the bloodstream infection15; (2) attended a hospital or hemodialysis clinic or received IV chemotherapy within 30 days before diagnosis of bacteremia; (3) was admitted to an acute care hospital for 2 or more days in the 90 days before diagnosis of bacteremia; or (4) resided in a nursing home or a long-term care facility. Cases of bacteremia were categorized as community-acquired if the first blood sample with positive culture result for P. aeruginosa was drawn within 48 h after hospital admission and the patient did not fit the criteria for a health care–associated infection.
Antimicrobial therapy was deemed appropriate if the cultured isolate possessed in vitro susceptibility to the antibiotic. Concomitant antipseudomonal therapy was defined as combination therapy if the non–ß-lactam antipseudomonal antibiotic was administered within 72 h of the initial ß-lactam antipseudomonal therapy and was continued for at least 72 h in conjunction with ß-lactam therapy. A body site was deemed to be the source of infection if it was documented as such in the patient’s chart or if P. aeruginosa was isolated from the site within 72 h of the incident blood sampling for culture. If P. aeruginosa was not isolated from samples of clinical material and clinical evidence of the source of bacteremia was lacking, the source was considered unknown. An isolate was considered multidrug-resistant if it was resistant to at least 1 agent in 3 or more antipseudomonal antibiotic categories (i.e., antipseudomonal penicillin, cephalosporin, carbapenem, fluoroquinolone, or aminoglycoside).16
The dosing regimens for ß-lactam antibiotics were prescribed at the physician’s discretion. According to institutional recommendations, the standard antibiotic doses used during the study period for treating P. aeruginosa bacteremia were ceftazidime 2 g IV q8h infused over 15–30 min, meropenem 500 mg IV q6h infused over 15–30 min (or, for central nervous system infection, 2 g IV q8h infused over 15–30 min), and piperacillin–tazobactam 4.5 g IV q6h infused over 30–60 min, each with dose adjustment for patients with calculated creatinine clearance of ≤ 30 mL/min.
The primary endpoints of this study were clinical and microbiological outcomes and costs (both total hospital costs and infection-related costs). Clinical response was evaluated at the end of therapy as clinical success or failure. Clinical success was defined as documented resolution of bacteremia and/or resolution of all signs and symptoms of infection. Patients with documentation of resolving bacteremia and transition to oral antipseudomonal antimicrobial therapy or discharge on IV antipseudomonal ß-lactam therapy were categorized as having clinical success. Patients with no response to initial antipseudomonal ß-lactam therapy and requiring a change in antibiotics were categorized as having clinical failure. Patients with transition to oral antipseudomonal antimicrobial therapy or discharge on IV antipseudomonal ß-lactam therapy (but without documentation of resolving bacteremia) were categorized as having clinical improvement. Microbiological response was evaluated only for living patients with repeat blood culture within 14 days after the end of therapy, where cure was defined as a documented negative blood culture result within 14 days after the end of antipseudomonal ß-lactam therapy. Mortality was defined as death from any cause while receiving therapy or within 14 days and 30 days after the end of antipseudomonal ß-lactam therapy.
Patient-specific costs of the hospital stay (total and categorized by hospital service) were obtained for each patient for both the total hospital stay and the infection-related hospital stay from the Business Office at Sunnybrook Health Sciences Centre. Infection-related hospital stay was defined as the time from the earlier of day of sampling for culture or start of ß-lactam therapy until the earlier of discharge or 7 days after cessation of ß-lactam therapy. Case costing captured both direct patient care costs and indirect overhead costs incurred during a patient’s stay. The costing methodology utilized the hospital’s financial data and distributed the costs on the basis of patient-specific utilization data. Workload minutes formed the basis for relative value units (RVUs), which measured the amount of resources required to complete a specific task for each individual patient.17 For each department, the direct and indirect costs could then be calculated per RVU, by dividing the total expense by the total RVUs. For certain departments, such as the operating room, data concerning specific supplies used by each patient were collected and actual supply costs applied (in addition to workload units). All cost data refer to actual costs at the time of service.
Results are expressed as mean ± standard deviation (SD) (and range), median (and range), or proportion of the total number of patients, depending on whether data were interval and parametric, interval and nonparametric or ordinal, or nominal, respectively. GraphPad InStat (version 3.05, 32-bit for Win95/NT; GraphPad Software Inc, La Jolla, California) was used to test data for normality using the Kolmogorov–Smirnov test. For comparisons among the antipseudomonal ß-lactam antibiotics (ceftazidime, meropenem, and piperacillin–tazobactam), one-way analysis of variance (ANOVA) was used for interval data that were normally distributed and had equal SDs, with the Tukey post-test being used for multiple comparisons when the p value from the ANOVA was less than 0.05; the Kruskal–Wallis test was used for interval data that were not normally distributed and/or had unequal SDs, with the Dunn post-test being used for multiple comparisons when the p value from the Kruskal–Wallis test was less than 0.05; and the χ2 test for proportions was used for nominal data, with the Marascuilo post-test procedure being used for multiple comparisons when the p value from the χ2 test for proportions was less than 0.05. Mortality rates (at 14 and 30 days) and rates of clinical cure and microbiological cure based on the prescribed antipseudomonal ß-lactams (ceftazidime, meropenem, and piperacillin–tazobactam) were compared by χ2 for proportions (Microsoft Office Excel 2007, Microsoft Corporation, Redmond, Washington) for initial screening (p < 0.05 defined as significant) and then confirmed using the Pearson correlation analysis (p < 0.05) followed by iterative binary logistic regression inputting all covariates with p < 0.05 to identify the best regression model (i.e., the model with the lowest p value, with a maximum p < 0.05 considered statistically significant and containing only variables that remained statistically significant on their own). Infection-related costs were compared by site of acquisition of P. aeruginosa bacteremia (nosocomial versus health care–associated) using an unpaired t test when the test for normality was passed and SDs were not significantly different (F test, p ≥ 0.05), an unpaired t test with Welch correction when the test for normality was passed and SDs were significantly different (F test, p < 0.05), or a Mann–Whitney U test when the test for normality failed. Standard sample-size equations for dichotomous data were used to determine the sample size required to identify a difference of at least 10 percentage points18 in mortality rate at 30 days (2-tailed, p = 0.05, power 0.80) in a prospective comparative study.
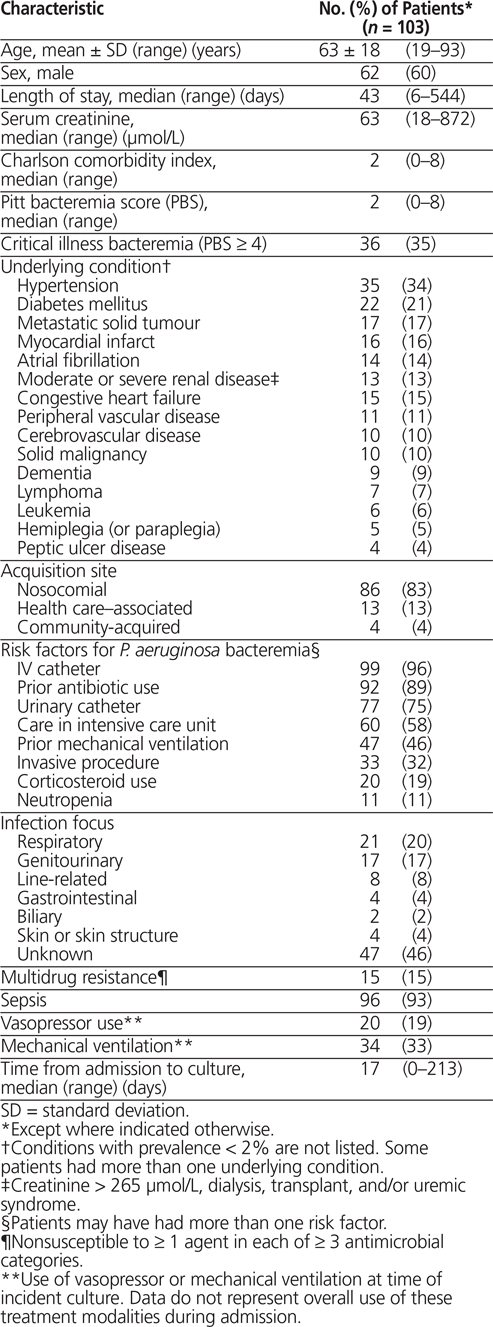
A total of 103 patients meeting the inclusion criteria were identified between March 1, 2005, and March 31, 2013. The mean age (± SD) was 63 ± 18 years (range 19–93 years), and 60% (62/103) were men (Table 1). About one-third of the patients (36/103 [35%]) were critically ill, defined as having a Pitt bacteremia score of at least 4.13,19,20 The majority of cases of P. aeruginosa bacteremia were acquired nosocomially (86/103 [83%]). The length of stay in hospital before the first positive blood culture result ranged from 0 to 213 days (median 17 days). A total of 60 patients (58%) were receiving care in the intensive care unit at the time bacteremia was diagnosed. On the day of sampling for culture, 96 patients (93%) met the criteria for sepsis.21
Table 1. Patient Characteristics in a Study of Pseudomonas aeruginosa Bacteremia
Of the 103 patients, 35 (34%) received ceftazidime, 20 (19%) received meropenem, and 48 (47%) received piperacillin–tazobactam. The median time from sample collection to initiation of ß-lactam therapy was 2 days (range −3 to 6 days), and the median duration of ß-lactam therapy was 8 days (range 3 to 45 days). Concomitant non–ß-lactam antipseudomonal therapy was prescribed for 26 patients (25%), with aminoglycosides being the most commonly used concomitant therapy (13/26 [50%]), followed by ciprofloxacin (10/26 [38%]). Ten patients (10%) were started on double antipseudomonal antibiotic coverage from the onset of therapy. Three patients (3%) received dual ß-lactam antipseudomonal agents: for 2 of these patients, the agents were prescribed for the purpose of double antipseudomonal coverage, and for 1 patient the combination was used for concomitant Stenotrophomonas maltophilia pneumonia. Susceptibilities of P. aeruginosa to the antipseudomonal ß-lactams were 85% (88/103), 79% (81/102), and 91% (94/103) for ceftazidime, meropenem, and piperacillin–tazobactam, respectively. Multidrug-resistant isolates were cultured in 15/103 (15%) of the positive isolates, and 96/103 patients (93%) received appropriate ß-lactam antipseudomonal therapy according to culture and susceptibility profiles.
A total of 6 (6%) patients died while receiving antipseudomonal ß-lactam antibiotic therapy; in addition, 17 (17%) patients died within 14 days and 28 patients (27%) within 30 days of discontinuation of antipseudomonal ß-lactam antibiotic therapy. At the end of therapy, 79 patients (77%) were deemed to have a clinical cure. Reasons for changing the initial antipseudomonal ß-lactam antibiotic therapy despite documented clinical cure included patient’s experience of an adverse effect, susceptibility results suggesting intermediate susceptibility or resistance, transition to an oral antibiotic, narrowing of the spectrum of antimicrobial coverage to minimize the use of broad-spectrum antibiotics, and treatment of concomitant infections that were unlikely to be susceptible to the initial agent.
Of the 47 patients with evaluable microbiological data, 41 (87%) were determined to have microbiological cure. No differences in clinical success rates were detected among the individual ß-lactam agents; however, the microbiological cure rate for piperacillin–tazobactam was 25% lower than that of both ceftazidime and meropenem (p = 0.037) (Table 2). This preliminary analysis was confirmed with a statistically significant model using binary logistic regression (p < 0.001), where the outcomes with ceftazidime and meropenem were pooled and compared with piperacillin–tazobactam as the reference. The microbiological response with piperacillin–tazobactam was significantly lower even though the time from sampling for culture to initiation of antipseudomonal ß-lactam therapy was significantly longer with ceftazidime than with piperacillin–tazobactam (2.3 versus 0.87 days, p < 0.001) and the percentage of patients with P. aeruginosa sensitive to meropenem was significantly lower than the percentage with P. aeruginosa sensitive to either ceftazidime or piperacillin–tazobactam (p < 0.05) (Table 2). Other parameters that remained in the best binary logistic regression model were length of stay and maximum temperature, both of which were negatively correlated with microbiological outcome.
Table 2. Outcomes by Antipseudomonal Agent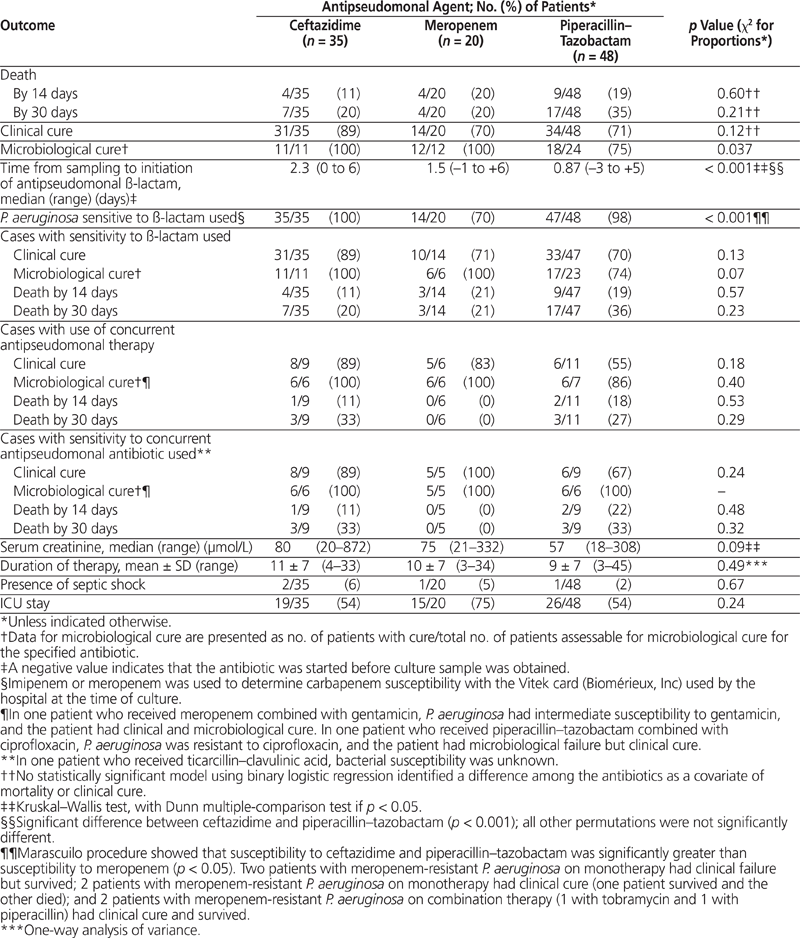
A total of 91 patients were included in the analysis of costs of the hospital stay, as costing data were available only for the period from April 2007 to December 2012. The median costs of the hospital stay and the infection-related stay for a patient with P. aeruginosa bacteremia were $121 718 and $29 697, respectively (Table 3). The number of patients with costing data for community-acquired P. aeruginosa bacteremia was too small (n = 3) for inclusion in any statistical analysis. Patients with nosocomial P. aeruginosa bacteremia had significantly higher costs for both total hospital stay and infection-related stay than patients with health care–associated P. aeruginosa bacteremia (p < 0.001) (Table 4).
Table 3. Costs Associated with Hospital Stay*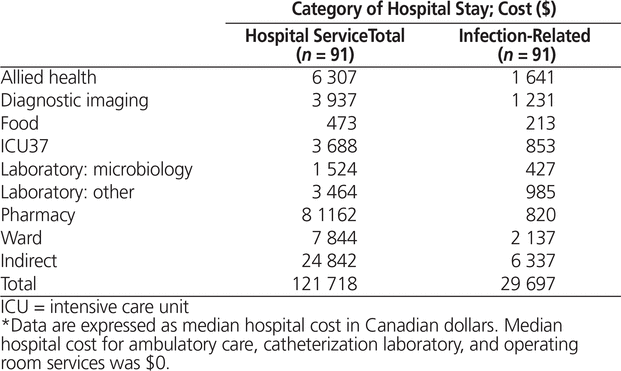
Table 4. Costs of Hospital Stay by Site of Acquisition of Infection*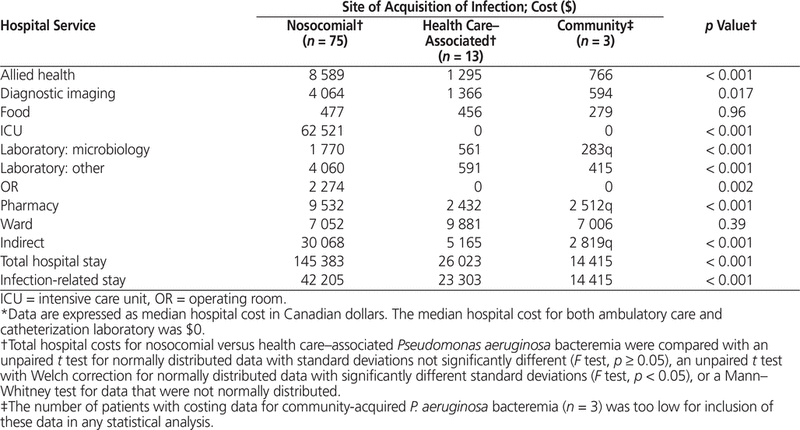
About 290 patients per group would be needed to perform a clinical trial designed to detect a 30-day mortality difference of at least 10 percentage points (that is, a decrease in mortality rate from 27% to 17%) between intermittent- and extended-infusion ß-lactam therapy (2-tailed test, p = 0.05, and 80% power).
In a population of patients with primarily nosocomial P. aeruginosa bacteremia, this study identified a 30-day non-attributable mortality rate of 27% for patients treated with antipseudomonal ß-lactam therapy administered by intermittent infusion. There was no difference among the ß-lactams evaluated (ceftazidime, meropenem, and piperacillin–tazobactam) in terms of mortality or clinical cure.
However, piperacillin–tazobactam administered at 4.5 g IV q6h, adjusted for renal function, was associated with a significantly worse microbiological outcome (χ2 analysis, p = 0.037; significant binary logistic model, p < 0.001). The median costs of hospital stay and infection-related stay for an inpatient with P. aeruginosa bacteremia were $121 718 and $29 697, respectively, and costs were significantly higher for patients with nosocomial P. aeruginosa bacteremia than for patients with health care– associated bacteremia.
For patients with potentially severe infections, such as P. aeruginosa, the administration of appropriate early empiric antibiotic therapies at doses appropriate to renal function can improve clinical outcomes, including survival.5,22 In addition, consideration should be given to the administration of dosing regimens that optimize the pharmacokinetic and pharmacodynamic (PK/PD) killing properties of the antibiotic in vivo.23,24 The PK/PD properties of ß-lactam antibiotics are considered to be optimized when the time above the MIC is achieved for the greatest proportion of the dosing interval (%f T>MIC),23,24 and some clinical data exist to support this method of administration.18,25,26
The finding of lower microbiological cure with piperacillin– tazobactam relative to ceftazidime and meropenem is supported by the PK/PD findings of Zelenitsky and others,24 who identified that of all antibiotics evaluated, piperacillin–tazobactam was associated with the greatest difficulty in achieving higher %f T>MIC targets in the ICU patient population. These authors suggested the use of extended infusions of piperacillin–tazobactam to achieve PK/PD targets comparable to those of the other broad-spectrum ß-lactam antibiotics.24
To pursue a clinical trial comparing intermittent and extended infusion of antipseudomonal ß-lactams, about 290 patients per group would be needed to detect a difference of 10 percentage points in 30-day mortality, from 27% to 17% (2-tailed test, p = 0.05, and 80% power). At the study centre, a median of only 15 eligible patients were identified per year; therefore, a multicentre trial would be required to evaluate a difference in mortality between intermittent and extended infusion of broad-spectrum ß-lactam antibiotics for pseudomonal bacteremia. Given the significant amount of time and resources that would be required to perform this type of multicentre trial, we have decided that, for the study institution, it may be in the best interests of patient care to use the current published literature to justify implementation of extended-infusion ß-lactam therapy, especially for critically ill patients.18,26,27
To our knowledge, this is the first study to determine the costs of total hospital stay and infection-related stay for patients admitted with P. aeruginosa bacteremia. This information may be useful for benchmarking purposes in designing antimicrobial stewardship, infection prevention and control, and antibiotic administration initiatives (e.g., extended infusion, combination antipseudomonal therapy, early administration of antibiotics in patients with bacteremia).
This study had several limitations associated with its small sample size and descriptive, single-centre, retrospective design. The influence of bias in the clinical evaluation is indisputable. These results apply only to monomicrobial bacteremia with P. aeruginosa, since patients with polymicrobial bacteremia were excluded. Conversely, the advantage to this approach is that it generated outcome data (clinical, microbiological, and cost) that apply specifically to P. aeruginosa bacteremia. Patients who received less than 72 h of antipseudomonal ß-lactam therapy were excluded, to minimize the risk of biasing clinical and microbio-logical outcomes by including either patients in whom an infection was ruled out or patients in whom insufficient duration of therapy could confound outcomes in a retrospective study where reasons for shortened therapy could not be determined. However, doing so introduced a selection bias for not capturing patients with true P. aeruginosa bacteremia who died from their infection within 72 h of initiation of ß-lactam antipseudomonal antibiotic therapy. This study was not designed to make direct comparisons between antibiotics, and although it had sufficient sample size to compare microbiological outcome between antibiotics, it was not sufficiently powered to compare clinical cure or mortality. In addition, definitive conclusions cannot be drawn regarding the findings for microbiological outcomes, because the retrospective design precluded evaluation of confounding factors that might have affected the results (e.g., underlying diseases, severity of illness, source of bacteremia, P. aeruginosa MIC). Costing data were available for patients admitted between April 2007 and December 2012 (91 of the 103 patients in the study). These costing data were based on the hospital’s financial data, which determine utilization data specific to the time of admission. This approach may underestimate the current costs of treating P. aeruginosa bacteremia.
Despite its limitations, this study provides important information about clinical, microbiological, and cost outcomes related to monomicrobial bacteremia caused by P. aeruginosa and treated with intermittent infusions of antipseudomonal ß-lactam antibiotics. To the best of the authors’ knowledge, this study is the first to identify total and infection-related hospital costs for patients with P. aeruginosa bacteremia and to characterize the cost differences for hospital-acquired versus health care– associated and community-acquired P. aeruginosa bacteremia. Finally, the study design allowed estimation of sample size for the conduct of a prospective comparative study of intermittent versus extended infusion of antipseudomonal ß-lactam antibiotics in the setting of P. aeruginosa bacteremia, which may be useful in the design of future investigations.
1. Walkty A, Baxter M, Adam H, Karlowsky JA, Lagacé-Wiens P, Hoban DJ, et al. Antimicrobial susceptibility of Pseudomonas aeruginosa isolates obtained from patients in Canadian hospitals: CANWARD 2008–2011. Diagn Microbiol Infect Dis. 2012;73(4):361–4.
2. Zavascki AP, Carvalhaes CG, Picão RC, Gales AC. Multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii: resistance mechanisms and implications for therapy. Expert Rev Anti Infect Ther. 2010; 8(1):71–93.
3. Richards MJ, Edwards JR, Culver DH, Gaynes RP. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit Care Med. 1999;27(5):887–92.
4. Gallagher PA, Watanakunakorn C. Pseudomonas bacteremia in a community teaching hospital, 1980–1984. Rev Infect Dis. 1989;11(6):846–52.
5. Morata L, Cobos-Trigueros N, Martinez JA, Soriano A, Almela M, Marco F, et al. Influence of multidrug resistance and appropriate empirical therapy on the 30-day mortality rate of Pseudomonas aeruginosa bacteremia. Antimicrob Agents Chemother. 2012;56(9):4833–7.

6. Aliaga L, Mediavilla JD, Cobo F. A clinical index predicting mortality with Pseudomonas aeruginosa bacteremia. J Med Microbiol. 2002;51(7):615–9.
7. Al-Hasan MN, Wilson JW, Lahr BD, Eckel-Passow JE, Baddour LM. Incidence of Pseudomonas aeruginosa bacteremia: a population-based study. Am J Med. 2008;121(8):702–8.

8. Craig W. Interrelationship between pharmacokinetics and pharmacodynamics in determining dosage regimens for broad-spectrum cephalosporins. Diagn Microbiol Infect Dis. 1995;22(1–2):89–96.
9. Roberts JA, Paul SK, Akova M, Bassetti M, De Waele JJ, Dimopoulos G, et al. DALI: defining antibiotic levels in intensive care unit patients: are current beta-lactam antibiotic doses sufficient for critically ill patients? Clin Infect Dis. 2014;58(8):1072–83.
10. Dellit TH, Owens RC, McGowan JE Jr, Gerding DN, Weinstein RA, Burke JP, et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis. 2007; 44(2):159–77.
11. Tamma PD, Putcha N, Suh YD, Van Arendonk KJ, Rinke ML. Does prolonged ß-lactam infusions improve clinical outcomes compared to intermittent infusions? A meta-analysis and systematic review of randomized, controlled trials. BMC Infect Dis. 2011;11:181.
12. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbitidy in longitudinal studies: development and validation. J Chron Dis. 1987;40(5):373–83.
13. Chow JW, Yu VL. Combination antibiotic therapy versus monotherapy for gram-negative bacteraemia: a commentary. Int J Antimicrob Agents. 1999; 11(1):7–12.
14. Friedman ND, Kaye KS, Stout JE, McGarry SA, Trivette SL, Briggs JP, et al. Health care–associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med. 2002;137(10):791–7.
15. Hall RG 2nd, Giuliano CA, Haase KK, Hazlewood KA, Frei CR, Forcade NA, et al. Empiric guideline-recommended weight-based vancomycin dosing and mortality in methicillin-resistant Staphylococcus aureus bacteremia: a retrospective cohort study. BMC Infect Dis. 2012;12:104.

16. Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–81.
17. Dummit L. The basics—relative value units (RVUs). Washington (DC): George Washington University; 2009.
18. Dulhunty JM, Roberts JA, Davis JS, Webb SA, Bellomo R, Gomersall C, et al. Continuous infusion of beta-lactam antibiotics in severe sepsis: a multicenter double-blind, randomized controlled trial. Clin Infect Dis. 2013;56(2):236–44.
19. Hill PC, Birch M, Chambers S, Drinkovic D, Ellis-Pegler RB, Everts R, et al. Prospective study of 424 cases of Staphylococcus aureus bacteraemia: determination of factors affecting incidence and mortality. Intern Med J. 2001;31(2):97–103.
20. Rhee JY, Kwon KT, Ki HK, Shin SY, Jung DS, Chung DR, et al. Scoring systems for prediction of mortality in patients with intensive care unit-acquired sepsis: a comparison of the Pitt bacteremia score and the Acute Physiology and Chronic Health Evaluation II scoring systems. Shock. 2009;31(2):146–50.
21. Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 1992;101(6):1644–55.
22. Micek ST, Lloyd AE, Ritchie DJ, Reichley RM, Fraser VJ, Kollef MH. Pseudomonas aeruginosa bloodstream infection: importance of appropriate initial antimicrobial treatment. Antimicrob Agents Chemother. 2005;49 (4):1306–11.

23. McKinnon PS, Paladino JA, Schentag JJ. Evaluation of area under the inhibitory curve (AUIC) and time above the minimum inhibitory concentration (T>MIC) as predictors of outcome for cefepime and ceftazidime in serious bacterial infections. Int J Antimicrob Agents. 2008;31(4):345–51.
24. Zelenitsky SA, Ariano RE, Zhanel GG. Pharmacodynamics of empirical antibiotic monotherapies for an intensive care unit (ICU) population based on Canadian surveillance data. J Antimicrob Chemother. 2011;66(2):343–9.
25. Lodise TP Jr, Lomaestro B, Drusano GL. Piperacillin–tazobactam for Pseudomonas aeruginosa infection: clinical implications of an extended-infusion dosing strategy. Clin Infect Dis. 2007;44(3):357–63.
26. Falagas ME, Tansarli GS, Ikawa K, Vardakas KZ. Clinical outcomes with extended or continuous versus short-term intravenous infusion of carbapenems and piperacillin/tazobactam: a systematic review and meta-analysis. Clin Infect Dis. 2013;56(2):272–82.
27. Chytra I, Stepan M, Benes J, Pelnar P, Zidkova A, Bergerova T, et al. Clinical and microbiological efficacy of continuous versus intermittent application of meropenem in critically ill patients: a randomized open-label controlled trial. Crit Care. 2012;16(3):R113.

Competing interests: None declared.
The authors thank the Business Office of Sunny-brook Health Sciences Centre for assistance with collection of patient-specific costing data.
Funding: None received.
Canadian Journal of Hospital Pharmacy, VOLUME 68, NUMBER 5, September-October 2015