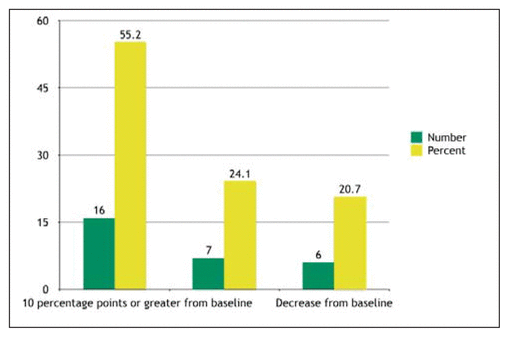
Figure 1 Changes in documentation by individual pharmacists, September 2016 to August 2017.
Stephanie Zimmer, Carolyn Gray, Caitlin Roy, William M Semchuk
Documentation in a patient’s medical record is an integral component of quality health care and as such is a legal requirement for health care providers. It is an avenue to communicate professional judgment, critical thinking, and plans for care among health care professionals; it also provides data to allow better understanding of care issues. Documentation in the medical record by pharmacists has the potential to minimize medication discrepancies and subsequent patient harm while forming a record of the level of care provided by the health care team.1 The importance of documentation by pharmacists is emphasized by the National Association of Pharmacy Regulatory Authorities (NAPRA) and the Canadian Society of Hospital Pharmacists (CSHP), which both include documentation as a professional practice standard in Canada.1,2
Although the importance of documentation is well known,3 pharmacists have reported that they often do not document in the medical record.4 Pullinger and Franklin4 surveyed 39 pharmacists and conducted a focus group with 32 clinical hospital pharmacists in London, England. Common barriers to adequate and complete documentation by pharmacists in the medical record included a preference for verbal communication, fear of criticism from prescribers, lack of belief in the significance of the intervention, and lack of ownership of the medical record. These authors suggested that hospitals develop a formal policy regarding pharmacist documentation and offer suitable training, including what and when to document. Herritt and others5 reported that the clinical activities most commonly documented by pharmacists were clarifications, order sets, clinical progress notes, and pharmacist suggestions. In contrast, in a more recent study, Baranski and others6 found that common topics for documentation included resolution of drug-related problems, pharmacokinetic consultation and recommendations, and patient education sessions.
Documentation of patient care issues by pharmacists was an expectation within the Regina Qu’Appelle Health Region, now known as the Saskatchewan Health Authority – Regina area (SHA–Regina). To better understand the quality and frequency of documentation within the local institutions, as well as alignment with CSHP guidelines, a residency project was conducted in 2015. The purpose of the project was to assess the competency of pharmacists in 18 elements of chart note documentation and to quantify the number of instances of documentation by pharmacists.6 The results showed that although pharmacists documented concisely, clearly, and in a diplomatic tone, there was room for improvement in the frequency and elements of chart note documentation in the medical record.
Further work to improve the frequency of documentation by pharmacists in SHA–Regina followed Kotter’s process for creating major change.7 With implementation of a collaborative prescribing agreement between pharmacists and physicians, the legal requirement for documentation was brought to the forefront of leaders’ minds. Legal counsel for the health region spoke to all staff pharmacists regarding the importance of complete and adequate documentation when prescribing medications. The urgency of the need to improve documentation led the clinical leadership team in SHA–Regina to establish a 3-year goal to increase both the quantity and quality of pharmacist documentation in the medical record.
To establish a baseline for documentation frequency, a survey of acute care clinical pharmacists was conducted at 2 tertiary care centres in Regina in September 2016. This group consisted of 35 pharmacists, of whom 40% had postbaccalaureate clinical training and 60% had more than 5 years of work experience. Of the 28 pharmacists who responded to the survey, 23 (82%) reported documenting fewer than half of their interventions in the progress notes. When asked why they did not document certain interventions, the majority of respondents (68% [19/28]) gave timing of the intervention as a reason (i.e., intervention performed during discussion on rounds or with the physician); a secondary reason was lack of time for documentation.
These survey results were compared with local metrics pertaining to documentation. Pharmacists are required to record their interventions on a daily basis using AIM-HIGH, a locally developed, Google Survey–based tracking tool. Tracking covers clinical pharmacy key performance indicators,8 as well as other key factors prioritized by the clinical leadership team, including documentation in the medical record. Data from the AIM-HIGH tool indicated that 18.96% of all interventions were documented in the progress notes between February and August 2016, which aligned with the self-reported results from the survey.
To gain a better understanding of documentation practices within the Canadian hospital pharmacy community, an environmental scan was conducted through the CSHP Pharmacy Specialty Networks (posted December 2016) and through e-mail contact with Saskatchewan hospital pharmacy managers. Six responses (from separate sites) were received. Respondents from 4 of the sites indicated that their pharmacists used a paper-based charting process as the primary means of documentation, whereas pharmacists at the other 2 sites used electronic medical records. At sites using paper-based charts, the progress note section was most commonly used (by 3 of the 4 sites). Training modules, templates, and policies and procedures were infrequently employed. These responses indicated a wide variety of documentation practices. In the SHA–Regina area, the medical record consists of a mixed paper and electronic system; pharmacists document in the progress note section of the paper chart, because they do not yet have access for documentation in the electronic record. A general SHA–Regina policy outlining the legal importance and logistics of documentation exists; however, activities that require documentation are left to the individual pharmacist’s discretion, and although educational certification regarding documentation exists, it is not mandated.
The Documentation Working Group (DWG) was formed in November 2016, with the specified goal of increasing, by 10 percentage points annually for 3 consecutive years, written documentation of acute care pharmacists’ interventions in progress notes, both at the individual pharmacist level and departmentally. The DWG also aimed to assess pharmacists’ satisfaction with the process used for improving documentation. Following a call for volunteers, initial DWG membership consisted of 5 clinical practice leaders (including C.G., C.R., W.M.S.) and 7 clinical acute care pharmacists from various specialties, which provided a representative sample of the staff pharmacists. The composition of the working group has been fluid, to account for departmental changes and to allow participation by a variety of staff members. The inclusion of front-line pharmacists was intended as a way to develop shared leadership and ownership of the project and to aid in implementation of interventions applicable to practice.
The DWG initially sought to identify local barriers that might be limiting documentation by pharmacists. These barriers were identified through brainstorming sessions within the DWG and informal feedback from other pharmacists. Proposed barriers included perceived lack of time, lack of clarity about what to document, and the perception that notes are not read by other health care professionals, especially physicians. Because the organization uses a paper-based medical record, further barriers included limited access to the medical record, such as during multidisciplinary rounds or when not present on the ward.
The DWG developed enablers to specifically overcome these identified barriers. To improve clarity about what was to be documented by pharmacists, the work standard “Pharmacist Documentation in Patient Progress Notes” (Appendix 1, available at https://www.cjhp-online.ca/index.php/cjhp/issue/view/189/showToc) was developed, outlining medication interventions that require documentation. A second work standard, “Medication Education Provided by Pharmacists” (Appendix 2, available at https://www.cjhp-online.ca/index.php/cjhp/issue/view/189/showToc), was developed to reduce repetitive documentation and improve efficiency. This work standard lists topics typically discussed with a patient during medication education counselling. When documenting an educational intervention, the pharmacist cites the standard, with the remainder of the documentation focused on identifying any components that were not addressed, any other relevant information that was discussed, and any patient concerns identified. This work standard was developed to address the barrier of lack of time, by reducing the extent of repetitive, non–patient-specific documentation required. Pharmacy leaders shared these work standards with care providers, and the standards themselves are available for reference by all health care professionals in SHA–Regina. Finally, a quality improvement assessment tool (Appendix 3, available at https://www.cjhp-online.ca/index.php/cjhp/issue/view/189/showToc) was developed using a template created during the previously noted residency project, to encourage pharmacists’ self-assessment of their notes, to improve the clarity of expected content for notes, and to increase confidence. As each enabler was developed, all pharmacists were educated on its use and were given the opportunity to provide feedback before departmental implementation.
To facilitate ongoing improvement and awareness, the clinical pharmacists in the DWG acted as liaisons with their respective teams. They educated their colleagues on the tools and progress of the DWG, and provided regular feedback to the DWG. These processes reinforced the involvement of all front-line staff to ensure the documentation interventions were relevant and realistic.
Achievement of the DWG’s goals was evaluated using data collected through the AIM-HIGH system and a satisfaction survey of front-line pharmacists.
AIM-HIGH data from September 2016 to August 2017 showed an increase in documentation of 9.45 percentage points (from 18.96% to 28.41%) at the departmental level, or a 49.8% improvement in the rate of documentation. All pharmacists had exposure to the interventions, and the 29 pharmacists who were practising at both baseline and the 1-year point were also assessed individually; 16 (55%) of these pharmacists achieved an increase of at least 10 percentage points in documentation rate (Figure 1). Individual assessments were completed to provide auditing and feedback for motivation. These results demonstrated an improvement by both individual pharmacists and the department as a whole and represent the first results of a 3-year, ongoing strategy. Documentation results were shared with all acute care pharmacists on a quarterly basis to ensure engagement in meeting the targeted goals for documentation.
|
|
||
|
Figure 1 Changes in documentation by individual pharmacists, September 2016 to August 2017. |
||
The acute care pharmacist team was surveyed in March 2018 to determine the level of satisfaction with the process used by the DWG to improve documentation rates. All respondents indicated that the working group provided an opportunity for the pharmacist team to somewhat or fully have ownership of decisions and the direction of practice change. They felt that the working group allowed members to feel included in determining direction for the department, and reported that they would support a similar process in the future. All agreed that they had been able to make a meaningful contribution and that the outcomes of the group were valid; none of the respondents stated that they would not participate in a similar process again.
This initiative had some limitations. The data collection tool, AIM-HIGH, required pharmacists to input their own interventions and activities, and the data may therefore have been an incomplete representation of activities performed. Also, individual comparative data were collected only for pharmacists who were practising during the baseline period, which excluded pharmacists new to the department or returning from leave.
The DWG initiative within SHA–Regina was able to increase the quantity and emphasize the importance of documentation of interventions by pharmacists. Although the department did not quite achieve the initial, arbitrarily selected goal of a 10 percentage point increase, documentation by pharmacists improved by almost 50%. The benefits of increasing documentation include the potential for improved communication between pharmacists and other health care providers, as was anecdotally reported by front-line staff. Greater documentation of activities in the progress notes can reduce duplication of work by pharmacists providing subsequent care and can lead to timely implementation of care plans. Improved satisfaction and confidence were reported by staff through standardization of what needed to be documented in the medical record and how best to document it.
The activities chosen to improve documentation stemmed from identifying barriers that pharmacists encounter when performing documentation in practice. The barriers identified by our team were similar to those identified by Pullinger and Franklin4 and included a perceived lack of time, uncertainty about how to document in certain situations, and the perception that notes are not read by other health care professionals, especially physicians. SHA–Regina pharmacists also identified that the shared, paper-based medical record used within the organization was a barrier to timely access for documentation. Although the barriers identified in SHA–Regina were not unique, a process to overcome these barriers for pharmacists has not been documented in the literature, to the best of our knowledge. The tools and knowledge translation initiatives that were implemented here focused on overcoming these site-specific barriers, and the resulting improvement in quantity of documentation highlights the benefits of using these techniques to change practice.
Although some of the barriers have been addressed, there remains room for improvement. A transition to complete electronic medical records is anticipated, which will improve access to the chart and reduce the time required for documentation. To continue improving the quantity of documentation to meet clinical and legal requirements, clinical leadership has set further goals for the DWG, which include prioritizing documentation of all activities relating to level II prescriptive authority in the progress note and working toward documentation of all pharmacists’ patient care activities in the medical record (electronic or paper chart). As defined by the Saskatchewan College of Pharmacy Professionals, level II prescriptive authority describes the ability of a pharmacist to prescribe select medications on the basis of a collaborative agreement between pharmacists and practitioners in a public health care institution.9 The quality and legibility of notes continue to improve through use of the self-assessment tool and education.
We have highlighted the process that we used to improve documentation rates within our acute care pharmacist group. However, there is a paucity of research describing the effect of pharmacist documentation on patient outcomes. Future research should assess the impact of increased pharmacist documentation on patient outcomes.
Both CSHP and NAPRA stress the importance of documentation in the medical record.1,2 A process based in change management, with front-line staff engagement, to improve documentation of interventions in the progress notes section of a patient chart in SHA–Regina was associated with an increase in documentation (by 9.45 percentage points) over 1 year. Additional stakeholder engagement strategies are being applied to continue efforts toward achieving the 3-year goal.
1 Documentation of pharmacists’ activities in the health record: guidelines. Ottawa (ON): Canadian Society of Hospital Pharmacists; 2013.
2 Model standards of practice for Canadian pharmacists. Ottawa (ON): National Association of Pharmacy Regulatory Authorities; 2009.
3 ASHP Council on Professional Affairs. ASHP guidelines on documenting pharmaceutical care in patient medical records. Am J Health Syst Pharm. 2003;60(7):705–7.
4 Pullinger W, Franklin BD. Pharmacists’ documentation in patients’ hospital health records: issues and educational implications. Int J Pharm Pract. 2010; 18(2):108–15.
5 Herritt L, MacInnis M, Grimm N, Lee R, MacLean D, MacNeil T, et al. Analysis of current pharmacist documentation practices in a tertiary care hospital [abstract]. Can J Hosp Pharm. 2014;67(4):318.
6 Baranski B, Bolt J, Albers L, Siddiqui R, Bell A, Semchuk W. Development of a documentation rubric and assessment of pharmacists’ competency for documentation in the patient health record. Can J Hosp Pharm. 2017;70(6): 423–9.
7 Kotter JP. Leading change. Boston (MA): Harvard Business School Press; 1996.
8 Fernandes O, Gorman SK, Slavik RS, Semchuk WM, Shalansky S, Bussières JF, et al. Development of clinical pharmacy key performance indicators for hospital pharmacists using a modified Delphi approach. Ann Pharmacother. 2015;49(6):656–69.
9 The regulatory bylaws of the Saskatchewan College of Pharmacy Professionals. Saskatoon (SK): Saskatchewan College of Pharmacy Professionals; 2018.
Competing interests: None declared. ( Return to Text )
Funding: None received. ( Return to Text )
Canadian Journal of Hospital Pharmacy, VOLUME 72, NUMBER 2, March-April 2019