Duane Bates, Sandy Zhang, Ben J Wilson, Shyane Wiegers
Aseptic meningitis is a clinical syndrome of meningeal inflammation in which common bacterial causes cannot be identified. The majority of cases are caused by viral infections, with enteroviruses being the most common organisms.1 Aseptic meningitis may also be caused by medications1,2 and has been most frequently reported with nonsteroidal anti-inflammatory drugs (NSAIDs; most commonly in association with ibuprofen), antibiotics (most commonly in association with trimethoprim, with or without sulfamethoxazole), anticonvulsants (most commonly in association with lamotrigine), and monoclonal antibodies.2,3 The interval between initiation of the medication and development of meningeal symptoms has ranged from minutes to 5 months.3 Patients typically present with fever, headache, photophobia, and neck stiffness. Other symptoms may include nausea, vomiting, diarrhea, phonophobia, arthralgia, myalgia, rash, or flu-like symptoms.1,2,4 Lumbar puncture for cerebrospinal fluid (CSF) analysis is required to confirm the diagnosis. CSF pleocytosis is nearly always present, and an increase in protein and a decrease in glucose, if present, are typically mild.1,2 Treatment of medication-associated aseptic meningitis involves stopping the offending agent and supportive care. Symptoms typically resolve within 5 days of drug discontinuation.2,3 We present a case of sulfasalazine-induced aseptic meningitis.
A 39-year-old woman presented to the emergency department with a 10-day history of malaise, nausea, and headache, which were associated with ocular pain, photophobia, tinnitus, and fever.* Past medical history included juvenile idiopathic arthritis, asthma, and previous upper gastrointestinal bleeding. The patient did not have a history of headaches or migraines. The patient’s home medications included budesonide 200 μg/formoterol 6 μg 2 puffs inhaled 1 or 2 times daily, salbutamol 100 μg 2 puffs inhaled as needed, melatonin 5 mg PO at bedtime as needed, ibuprofen 200 mg PO as needed, and cetirizine 10 mg PO daily as needed. Two weeks before the admission, the patient had been given a prescription for sulfasalazine for her arthritis. The sulfasalazine was started at a dosage of 500 mg PO daily for 7 days and was to be increased by 500 mg daily every 7 days to a dosage of 1000 mg PO twice daily. Before admission, the sulfasalazine dosage was 500 mg PO twice daily. The patient had not taken any NSAIDs after starting the sulfasalazine. The patient reported no use of other over-the-counter medications, herbal medications, natural health products, food supplements, or traditional medicines. The patient had smoked 1 pack of cigarettes per day for 20 years, drank about 10 alcoholic beverages per week, and occasionally used cannabis.
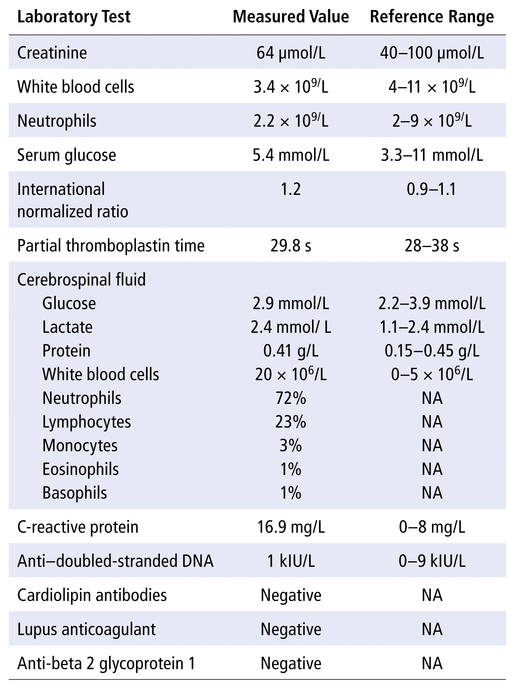
The patient was 159.8 cm tall and weighed 57.6 kg. Vital signs on admission were as follows: temperature 39°C, blood pressure 119/73 mm Hg, heart rate 108/min, and oxygen saturation 97% on room air. Physical examination showed no neck stiffness or deficit in range of motion. The Brudzinski sign, Kernig sign, and Jolt test were all negative. On neurological examination, motor and sensory functions were normal. Serum electrolytes, hemoglobin, platelets, and liver enzymes were normal. The CSF contained normal glucose, normal protein, and elevated white blood cells (20 × 106/L; normal range 0–5 × 106/L), including 72% neutrophils (Table 1). The results of urinalysis and chest radiography were unremarkable. The patient received 1 dose of ketorolac 10 mg IV and 1 dose of metoclopramide 10 mg IV for her headache, with no effect. Acetaminophen 1000 mg PO 4 times daily was started for her headache. In addition, the following medications were started: ceftriaxone 2 g IV every 12 hours, dexamethasone 10 mg IV every 6 hours, and acyclovir 650 mg IV every 8 hours. The patient received 2 doses of oral morphine for her headache. The sulfasalazine was discontinued on admission.
TABLE 1 Summary of Laboratory Test Results

On day 2 of admission, the patient received 2 doses of oral hydromorphone for her headache. Negative results were obtained for the following investigations: urine, blood, and CSF culture; respiratory pathogen panel; nasopharyngeal swab for COVID-19, influenza A/B, and respiratory syncytial virus; alpha herpes virus panel; and enterovirus and parechovirus panel. The ceftriaxone, dexamethasone, and acyclovir were discontinued because the patient’s symptoms were thought to be due to aseptic meningitis secondary to sulfasalazine. The patient was discharged on day 2, as the headache had improved and all other symptoms had resolved. The patient was advised to avoid the use of NSAIDs and to use only acetaminophen for headache and minor aches and pains. The headache had completely resolved 2 days after discharge. At 29 days after discharge, the patient had no recurrence of symptoms.
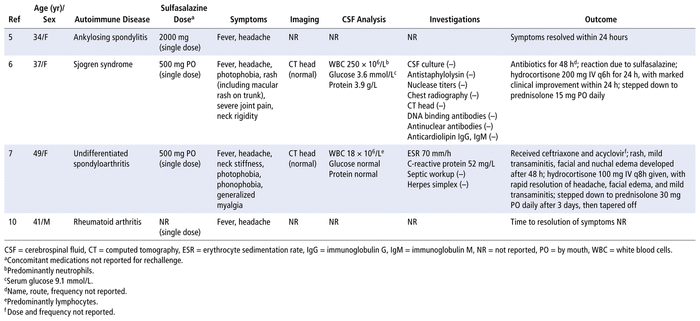
A literature search of PubMed, Google Scholar, and Embase databases from inception to March 30, 2021, with the search terms “aseptic meningitis” and “sulfasalazine” yielded 6 citations involving 7 patients5–10 (summarized in Table 2). The patients were predominantly female, with ages ranging from 34 to 74 years. All of the patients had an autoimmune disease. Fever, headache, vomiting, and neck stiffness were the most commonly reported symptoms. The onset of symptoms ranged from 12 days to a few months after exposure to the medication. Resolution of symptoms occurred 2 to 8 days after sulfasalazine discontinuation. Other infectious causes were ruled out in 6 of the 7 cases. Four patients were rechallenged and experienced recurrence of symptoms within 1 to 12 hours after a single dose of sulfasalazine (Table 3). Our patient had a similar presentation and temporal association, with clinical onset and resolution within the described time frames after sulfasalazine initiation and discontinuation, respectively.
TABLE 2 Summary of Case Reports of Sulfasalazine-Induced Aseptic Meningitis
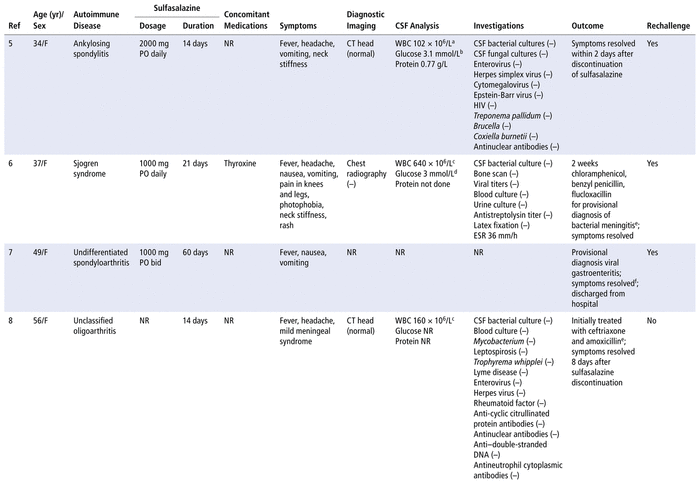
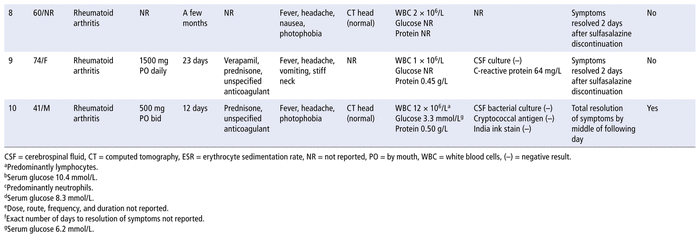
TABLE 3 Summary of Sulfasalazine Rechallenge
Two weeks after starting sulfasalazine, the patient described here presented with meningeal symptoms, and the results of work-up for infectious causes of meningitis were negative. The patient was not tested for other causes of viral induced aseptic meningitis, but these could be excluded on a clinical basis.1 She did not have risk factors for HIV, and testing for this virus was therefore not conducted. The patient had not been exposed to rodent excrement, and lymphocytic choriomeningitis virus was therefore ruled out. There had been no recent travel, so arthropod-borne flaviviruses, bunyaviruses, and orthobunyaviruses were excluded. She had been vaccinated against mumps and measles, which made these viruses unlikely as the cause of aseptic meningitis. The patient had not been bitten by an animal infected with rabies.
Neutrophilic pleocytosis of the CSF is a characteristic of drug-induced aseptic meningitis.2,3 Autoimmune diseases such as rheumatoid arthritis, systemic lupus erythematosus, Sjogren syndrome, and Behcet syndrome may predispose patients to development of aseptic meningitis.1,2 The patient in this case had juvenile idiopathic arthritis but did not report arthralgia, and her C-reactive protein was marginally elevated, which made active rheumatoid arthritis unlikely. Antiphospholipid antibody syndrome has been associated with migraines, but the results of work-up were negative.1 The rapid onset and resolution of signs and symptoms suggest that this was a case of aseptic meningitis secondary to sulfasalazine.2–4 The Naranjo Adverse Drug Reaction Probability Scale score was 6, suggesting a probable adverse reaction to sulfasalazine.11
Before admission, the patient had been taking ibuprofen as needed, but upon discharge she was advised to avoid NSAIDs. NSAIDs are the most frequent cause of drug-induced aseptic meningitis.2 Ibuprofen is the medication most commonly implicated, followed by diclofenac, naproxen, and sulindac.2,3 In a review of 72 cases of NSAID-induced aseptic meningitis, 62% of the cases involved females with a mean age of 39 years (standard deviation 15).3 Symptoms occurred within 30 minutes to 4 months (median 8 hours) of exposure.3 Risk factors for NSAID-induced aseptic meningitis in the case presented here include the patient’s sex, age, and history of juvenile idiopathic arthritis; upon discharge, she was therefore advised to use acetaminophen rather than NSAIDs.
Two mechanisms are thought to be involved in drug-induced aseptic meningitis. The first, related to direct toxicity of the drug, may involve circulating immune complex depositions in, or direct chemical irritation of, the meninges.2,4 The intrathecal route of administration increases the risk of meningitis, depending on the concentration of the drug, the molecular size, and the drug’s ability to cross lipid brain barriers.2 The second mechanism involves an immunological hypersensitivity that may be further subdivided into type I to IV hypersensitivity reactions.2,4 Type III and IV hypersensitivity reactions are the more likely mechanisms involved in drug-induced meningitis.2 In such reactions, antibodies combine to form complexes with the drug or its metabolite; these complexes activate complement (type III) or allow T cells reactive to the drug to be recruited to a site of inflammation (type IV).2 The exact mechanism by which sulfasalazine causes aseptic meningitis is unknown.
In the case reported here, the clinical syndrome and its close temporal relationship with sulfasalazine initiation and discontinuation are similar to other reported cases of sulfasalazine-induced aseptic meningitis. Infectious and rheumatic causes were satisfactorily excluded. Clinicians should consider a diagnosis of sulfasalazine-induced aseptic meningitis if other potential causes have been ruled out.
1 DynaMed [Internet]. EBSCO Information Services; 1995– [cited 2021 Apr 9]. Available from: http://www.www.dynamed.com. Registration and login required.
2 Yelehe-Okouma M, Czmil-Garon J, Pape E, Petitpain N, Gillet P. Drug-induced aseptic meningitis: a mini-review. Fundam Clin Pharmacol. 2018;32(3):252–60.
Crossref PubMed
3 Moris G, Garcia-Monco JC. The challenge of drug-induced aseptic meningitis revisited. JAMA Intern Med. 2014;174(9):1511–2.
Crossref PubMed
4 Kepa L, Oczko-Grzesik B, Stolarz W, Sobala-Szczygiel B. Drug-induced aseptic meningitis in suspected central nervous system infections. J Clin Neurosci. 2005;12(5):562–4.
Crossref PubMed
5 Salouage I, El Aidli S, Cherif F, Kastalli S, Zaiem A, Daghfous R. Sulfasalazine-induced aseptic meningitis with a positive rechallenge: a case report and review of the literature. Therapie. 2013;68(6):423–6.
Crossref PubMed
6 Merrin P, Williams IA. Meningitis associated with sulphasalazine in a patient with Sjogrens syndrome and polyarthritis. Ann Rhuem Dis. 1991;50(9):645–6.
Crossref
7 Tay SH, Lateef A, Cheung PP. Sulfasalazine-induced aseptic meningitis with facial and nuchal edema in a patient with spondyloarthritis. Int J Rheum Dis. 2012;15(4):e71–2.
Crossref PubMed
8 Houitte R, Abgueguen P, Masson C. Salazopyrine-induced aseptic meningitis. Bone Joint Spine. 2009;76(2):216–7.
Crossref
9 Gerster JC, Scherrer U, Biollaz J. Meningite aspetique sur salazopyrine: une oberservation et revue de la literature. Rhumatologie. 1998;50(6): 176–8.
10 Alloway JA, Mitchell SR. Sulfasalazine neurotoxicity: a report of aseptic meningitis and a review of the literature. J Rheumatol. 1993;20(2):409–11.
PubMed
11 Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239–45.
Crossref PubMed
Competing interests: None declared. ( Return to Text )
*The patient gave verbal consent for publication of this case report. ( Return to Text )
Funding: None received. ( Return to Text )
Canadian Journal of Hospital Pharmacy, VOLUME 75, NUMBER 4, Fall 2022